Hemocytometer Calculator
Calculate cell concentration, viability, dilution factor, and total cell counts from hemocytometer counts.
💡 Quick Summary
Calculate viable and dead cell concentrations, dilution factors, total cell counts, and viability percentages from hemocytometer counts. Uses the standard trypan blue exclusion method with the 10⁴ volume correction factor.
📋 How to Use
- Select the type of calculation from the I want to calculate dropdown.
- Enter the required values in the fields that appear.
- Click Calculate to see the result.
- Review the step-by-step calculation breakdown shown below the result.
- Click Reset to clear all fields and start again.
🧮 Formulas & Logic
📊 Result Interpretation
Expressed as cells/mL. Multiply by total culture volume (mL) to obtain the absolute cell count in your flask or tube.
A typical 1:1 trypan blue mix gives DF = 2. A 1:9 mix gives DF = 10. Always account for this in the concentration formula.
Excellent — culture is healthy and suitable for most experiments.
Acceptable — monitor closely and investigate potential stressors.
Poor — cells may be stressed, contaminated, or overgrown. Not recommended for sensitive assays.
Used to determine the number of cells to seed for passaging, seeding plates, or preparing samples.
🔬 Applications
- Cell culture viability assessment using trypan blue exclusion
- Determining cell seeding density before plating experiments
- Cell banking, cryopreservation, and thaw recovery assessment
- Drug cytotoxicity and cell death assays
- Primary cell isolation quality control
- Yeast cell counting in fermentation and brewing
- Proliferation and growth curve experiments
⚠️ Common Mistakes & Warnings
Trypan blue continues to enter cells over time. Count within 3–5 minutes of mixing to avoid overestimating dead cells and underestimating viability.
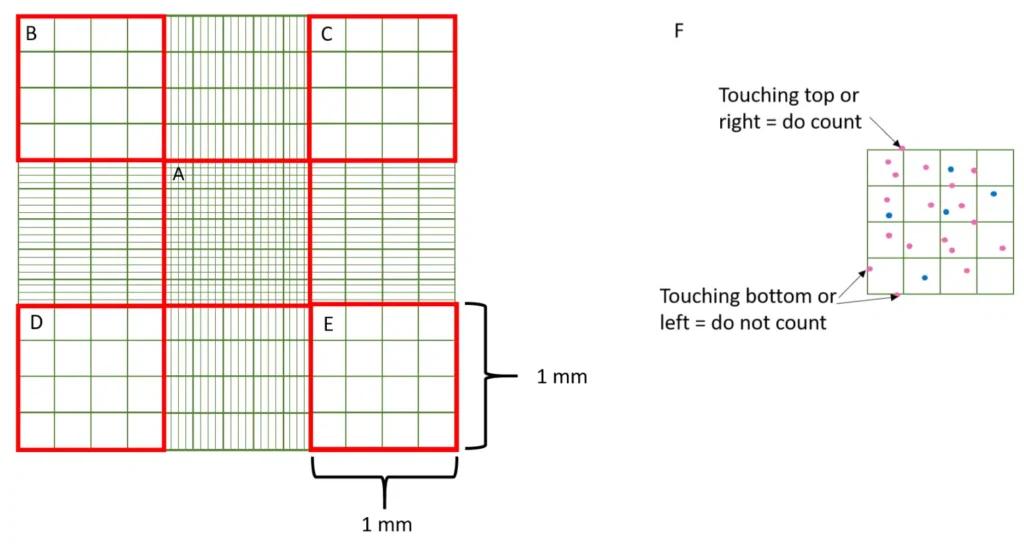
Count cells touching the top and left boundary lines of each square. Exclude cells touching the bottom and right lines. Consistent application of this rule across all counts minimises error.
Clumped cells are difficult to count accurately and skew results. Mix the cell suspension thoroughly before loading. If clumping persists, pipette gently several times or filter through a cell strainer.
Omitting the dilution factor (e.g., the trypan blue volume added) will underestimate cell concentration. A 1:1 mix of cells to trypan blue always gives a DF of at least 2.
For accurate counting, aim for 20–50 cells per large square. Fewer than 20 cells introduces high statistical error; more than 50 makes counting difficult and error-prone.
Count at least 4 large squares (or 100 total cells) to reduce counting variability. Averaging over more squares improves accuracy.
❓ Frequently Asked Questions
Why do we multiply by 10⁴?
What is trypan blue exclusion?
How many squares should I count?
What dilution factor should I use for trypan blue?
Why is my cell count inconsistent between chambers?
Can I use the hemocytometer for non-mammalian cells?
What is an acceptable viability for cell culture?
Concentration formula: (Cells ÷ Squares) × Dilution Factor × 10⁴ cells/mL. Based on standard hemocytometer chamber depth of 0.1 mm.
Counting cells during a regular passage or for experimental application:
1. Proceed based on whether the cell is suspended or adherent. Centrifuge cells at 220 xg for 5 min. Aspirate most of the supernatant, except for 100-200 μl to avoid disturbing pellet.
2. Re-suspend cell pellet in 1 mL medium, or more if large pellet, but keep track of what volume you use to re-suspend your cells.
3. Take 100 uL sample of re-suspended cells and add to Eppendorf tube.
4. Add 100 uL of Trypan blue to Eppendorf tube and pipette up and down a few times to ensure an even suspension. Gently pipette solution into each well of the hemocytometer in order to fill the area under the coverslip by capillary action.
(It is important not to disturb the coverslip in this process, to maintain an even distance between the glass and the hemocytometer grid. This ensures that each large square contains 0.0001 mL or 0.1 µL of suspension)
5. Count the number of dead and alive cells in 5 of the large squares of the hemocytometer grid, using phase contrast microscope under ~10x power.
(For standardization, count those in the 4 corner squares and center square. For cells touching an edge, count the ones touching the top and right edges and
exclude those touching bottom and left edges)
6. Dead cells appear dark/blue, as dye enters through cell membrane. Live cells appear more clear centrally.
Watch & Learn! 🎥 Check out the below ⬇️ video for a step-by-step guide on using a hemocytometer.
Try solving below: Question & Solution Inside! 🚀
Question: You are a researcher tasked with the setting up a series of experiments and analysing the resultant data from a study of the effects of the organophosphate chlorpyrifos on the viability and differentiation of cultured mouse N2a neuroblastoma cell line, as indicated below. You are required to handle, analyse, present and interpret the data provided, providing evidence of how you have arrived at your conclusions Cells were grown as a mitotic monolayer to approximately 80% confluence then detached for counting. After centrifugation of the resultant cell suspension, cell pellets were suspended in 1 mL growth medium. A volume of 10 µL of this cell suspension was mixed with 90 µL growth medium containing 0.2 % v/v Trypan Blue, after which the cells analysed in a haemocytometer, giving the data shown in Table 1.
| Square number* | Total cell count | Blue (dead) cell count |
| 1 | 99 | 3 |
| 2 | 115 | 8 |
| 3 | 87 | 6 |
| 4 | 105 | 5 |
| 5 | 125 | 9 |
*Each haemocytometer square has a volume of 10-4 ml. Using the data in Table 1, calculate the following parameters.
(i) Calculate total cell count/ml
(ii) Viable cell count/ml
(iii) % Viability
Solution:
Answer (i) Total cell count/mL means we have to calculate total concentration of live and dead cells.
Total cell count/mL = Average of total cells per square x dilution factor x 104
Average of total cells per square= Total cells counted ÷ Total number of squares counted
Average of total cells per square= (99+ 115 + 87 + 105 + 125) ÷ 5= 106.2
Dilution factor= 100 ÷ 10= 10
Total cell count/mL = 106.2 x 10 x 104 = 1.062 x 107 =10,620,000 cells/mL = 10.62 million cells/mL
Answer (ii) Viable cell count/mL = Average viable count per square x dilution factor x 104
For each square, calculate the number of viable cells = Total count – blue cell count
Average viable count per square = Total viable cells counted ÷ Total number of squares counted
Average viable count per square = (96+107+81+100+116) ÷ 5 = 100
Viable cell count/ml = Average viable count per square x dilution factor x 104
Viable cell count/ml = 100 x 10 x 104 = 1 x 10⁷ viable cells/mL = 10 million viable cells/mL
Answer (iii) % Viability= (Total number of viable cells per ml ÷ Total cell count per ml) x 100
% Viability= (10 million ÷ 10.62 million) x 100 = 94.2%
OR
% Viability= (Average viable count per square ÷ Average of total cells per square) x 100
% Viability= (100 ÷ 106.2) x 100 = 94.2%
Mathematical formulas
- Viable Cell Concentration (cells/ml) = Average number of viable cells per square x Dilution factor x 10⁴
If you are wondering 🤔 where did 10⁴ came from, here is the explanation below.
We multiply by 10,000 because the hemocytometer only allows us to count cells in a very small volume (0.1 µL or 0.0001 mL per large square). To express the concentration in a standard unit (cells per mL), we scale up by: 10,000. This accounts for the fact that there are 10,000 of these 0.0001 mL volumes in 1 mL, ensuring the final result represents the total number of cells per milliliter (0.0001ml x 10,000 = 1ml).

2. Dead Cell Concentration (cells/ml) = Average number of dead cells per square x Dilution factor x 10⁴
3. Dilution factor = Total volume after adding 0.2% trypan blue dye (µL) ÷ Initial volume of cell suspension (µL)
4. Total number of Live Cells in Stock Solution = Concentration of live cells (cells/ml) x Total volume of stock solution (ml)
5. Total number of Dead Cells in Stock Solution = Concentration of dead cells (cells/ml) x Total volume of stock solution (ml)
6. Percentage Cell Viability Calculator = (Number of Live Cells ÷ Total Number of Cells ) x100
7. Percentage Cell death Calculator = (Number of Dead Cells ÷ Total Number of Cells ) x100

